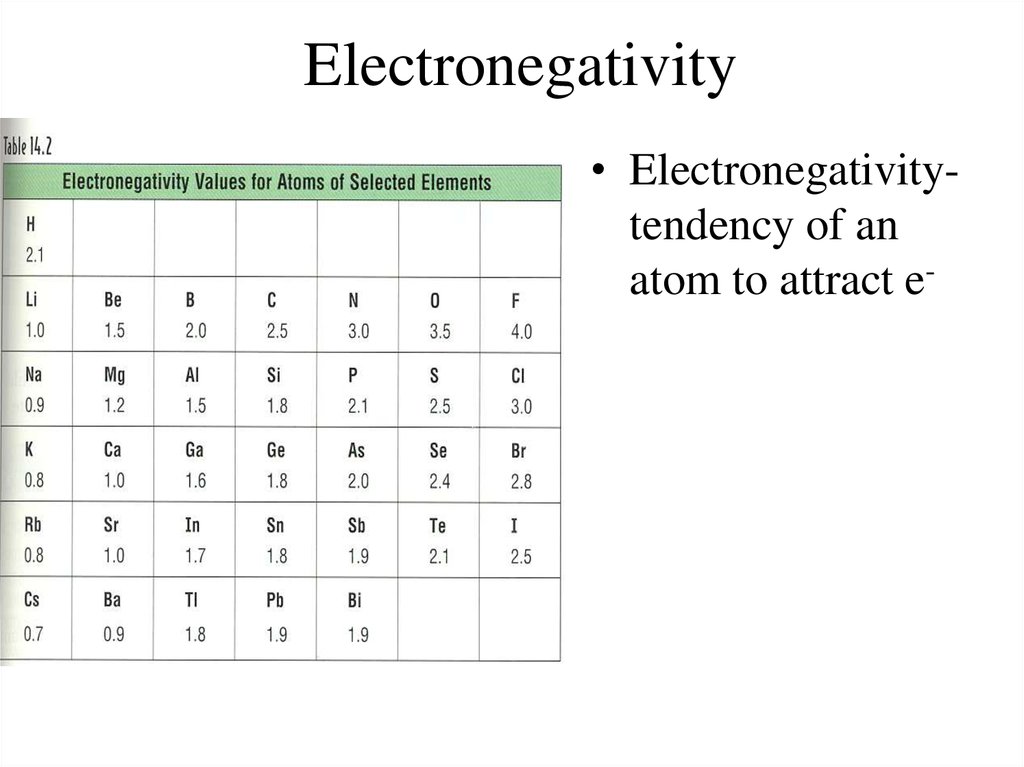
Metalloids are those seven elements that make up the stair-step line on the periodic table, including boron and silicon. There are only eight of them, but they can take on some very unique properties, charges, and colors.Īluminum (Al), tin (Sn), and lead (Pb) are post-transition metals. The post-transition metals are those metals that come after the transition metals, but before the noble gases within a row. Iron (Fe), platinum (Pt), molybdenum (Mo), nickel (Ni), palladium (Pd), zinc (Zn), and copper (Cu) are transition metals. As a result, these metals tend to have a wide variety of colors and properties. These metals are named this way, as they can take on many different charges, due to their valence electrons being in the d-orbitals. The transition metals include 34 metals in the middle of the periodic table. What are transition and post-transition metals? As an example, uranium (U) is a part of this group. These metals have very unique properties, owing to their f-orbital arrangements and unstable nuclei. The bottom row contains actinides, elements 89 – 103. The top row houses lanthanides, which are elements 57 – 70. Usually, they are shown as two rows on a separate section. The lanthanides and actinides refer to the two families of mostly radioactive metals in an off-shoot of the periodic table. They tend to form cations with a positive two charge and bond ionically. These metals are named for forming basic solutions when put into water. Its six elements extend from beryllium (Be) to radium (Ra). The alkaline earth metals represent the second column on the periodic table. Hydrogen (H) may be mistakenly placed into this family, but it does not contain any of the family traits. These elements form the ionic bonds known as salts. These metals all contain one valence electron, meaning they form cations and react readily with water.
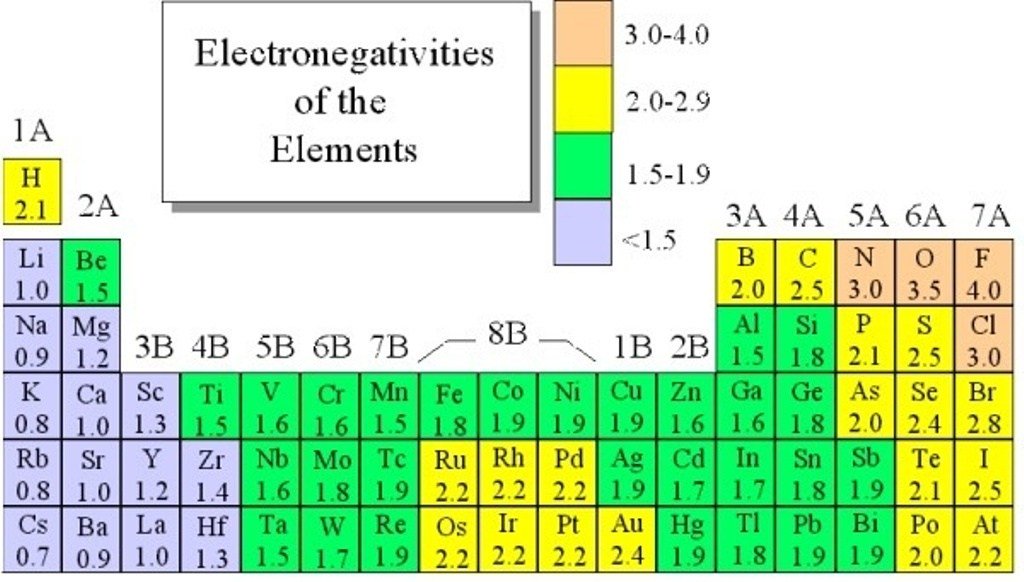
The alkali metal family includes the six elements in the first column of the periodic table, from lithium (Li) down to francium (Fr). For example, chromium (Cr) may be added to increase steel’s hardness. Its basic formula includes iron (Fe) with trace amounts of carbon (C).ĭifferent types of steel made add other elements to enhance properties. Only elements go onto the periodic table, and steel is an alloy, not an element. This means that they are not commonly found naturally on Earth or within the universe. Of those, the two most rare metals are rhodium (Rh) and osmium (Os). These metals are considered to be both very rare and of high value. On the periodic table, there is a family of eight elements known as the precious metals, including elements 44 – 47 like silver and 76 – 79 like gold. Odd-numbered elements pose a greater challenge for isolation, as their nucleons are not as stable. Those larger than element 109 have all been formed and isolated this way.Ĭurrently, the largest made is 118. However, heavier elements must be synthesized in a nuclear laboratory. Is there an element 119?Īs of this writing, scientists have not isolated a stable element 119. Here on Earth, elements like oxygen (O) and hydrogen (H) are common, but technetium (Tc) and nihonium (Nh) are rare. All of matter is made of these 118 elements, combined in various ratios and kept in different states, like solids.ĭepending on where you are in the universe, some are more prevalent. It consists of a specific number of protons and variable neutrons and electrons. What are the 118 elements?Īn element, simply put, is a fundamental unit of matter that cannot be easily broken down. Other information in the box includes the element’s electron configuration, electronegativity, oxidation states, and its first ionization energy measured in kilojoules per mole. The largest number is the element’s atomic number. Located right above the English name for each element, the largest letters represent its chemical abbreviation. We go in-depth on each of these groups in the questions below. The colored areas of our table break up the elements into groups of alkali metals, alkaline earth metals, lanthanides and actinides, transition and post-transition metals, metalloids, nonmetals, halogens, and noble gases. Some tables, like ours above, also include colors to help identify groups that cross rows and columns.

The table is comprised of seven rows (called periods), 18 columns (called groups or families). The key to understanding the periodic table is in its layout. This pattern is called a period (or row) on the periodic table. Based on the number of electrons, a scientist can determine which group to place an element into. When Dmitri Mendeleev drew up the periodic table, he grouped elements of similar properties. Here are some answers to common questions about the periodic table. Regulated Brokers: Where Can I Trade Metal Commodities?.

